中国组织工程研究 ›› 2013, Vol. 17 ›› Issue (18): 3349-3357.doi: 10.3969/j.issn.2095-4344.2013.18.017
• 细胞与组织移植 cell and tissue transplantation • 上一篇 下一篇
带血管肌筋膜包埋脂肪干细胞载体复合物构建血管化组织工程脂肪
王和庚1,黎洪棉1,崔世恩2,徐昆明1
- 1中山大学附属中山医院整形美容外科,广东省中山市 528403
2中山大学附属中山医院乳腺外科,广东省中山市 528403
-
收稿日期:2012-05-31修回日期:2012-07-24出版日期:2013-04-30发布日期:2013-04-30 -
通讯作者:黎洪棉,男,1971年生,2008年南方医科大学毕业,博士,主任医师,硕士生导师,主要从事整形美容外科和干细胞、组织工程与再生医学的研究。中山大学附属中山医院整形美容外科,广东省中山市 528403 -
作者简介:王和庚★,男,1972年生,硕士,副主任医师,主要从事烧伤整形临床与基础研究工作。 -
基金资助:课题受中国博士后科学基金(20090450910),广东省医学科研基金项目(A2011739)和中山市科技计划项目(20113A008)资助。
Vascularized tissue-engineered adipose established via the adipose-derived stem cells-attached scaffolds encapsulated in muscular fasciae with axial pattern blood vessel pedicle
Wang He-geng1, Li Hong-mian1, Cui Shi-en2, Xu Kun-ming1
- 1 Department of Plastic and Aesthetic Surgery, Postdoctoral Mobile Work Station, the Affiliated Zhongshan Hospital of Sun Yat-sen University, Zhongshan 528403, Guangdong Province, China
2 Department of Mammary Gland Surgery, the Affiliated Zhongshan Hospital of Sun Yat-sen University, Zhongshan 528403, Guangdong Province, China
-
Received:2012-05-31Revised:2012-07-24Online:2013-04-30Published:2013-04-30 -
Contact:Li Hong-mian, Doctor, Chief physician, Master’s supervisor, Department of Plastic and Aesthetic Surgery, Postdoctoral Mobile Work Station, the Affiliated Zhongshan Hospital of Sun Yat-Sen University, Zhongshan 528403, Guangdong Province, China binrong2112@163.com -
About author:Wang He-geng★, Master, Associate chief physician, Department of Plastic and Aesthetic Surgery, the Affiliated Zhongshan Hospital of Sun Yat-Sen University, Zhongshan 528403, Guangdong Province, China drwanghegeng@126.com -
Supported by:China Postdoctoral Science Foundation, No. 20090450910; Guangdong Medical Scientific Foundation, No. A2011739; Zhongshan Scientific and Technological Planning Projects, No. 20113A008
摘要:
背景:再血管化是构建组织工程化脂肪的关键因素。 目的:观察3种不同筋膜瓣包裹的脂肪来源干细胞与胶原蛋白支架复合物在体内成脂效率的差异。 方法:分离兔右侧带血管蒂的背阔肌筋膜瓣,包裹成脂诱导后的脂肪来源干细胞与Ⅰ型胶原蛋白复合物,设为带有轴型血管蒂的诱导分化组。分离兔左侧带血管蒂的背阔肌筋膜瓣,包裹未经诱导的脂肪来源干细胞与Ⅰ型胶原蛋白复合物,设为带有轴型血管蒂的未诱导分化组。分离兔右侧无特定血管蒂的臀大肌筋膜瓣,包裹未经诱导的脂肪来源干细胞与Ⅰ型胶原蛋白复合物,作为对照组。 结果与结论:移植后8周,苏木精-伊红染色和免疫组织化学染色检测结果显示,各组均可见新生脂肪组织形成,带有轴型血管蒂的诱导分化组新生肪组织平均湿质量和新生微血管密度均高于其他2组 (P < 0.01,P < 0.05)。结果说明实验成功构建了带血管肌筋膜包埋成脂诱导后的脂肪干细胞载体复合物构建血管化组织工程脂肪,此复合物在体内的成脂效率和促进血管再生的能力最好。
中图分类号:
引用本文
王和庚,黎洪棉,崔世恩,徐昆明 . 带血管肌筋膜包埋脂肪干细胞载体复合物构建血管化组织工程脂肪[J]. 中国组织工程研究, 2013, 17(18): 3349-3357.
Wang He-geng1, Li Hong-mian1, Cui Shi-en2, Xu Kun-ming1. Vascularized tissue-engineered adipose established via the adipose-derived stem cells-attached scaffolds encapsulated in muscular fasciae with axial pattern blood vessel pedicle[J]. Chinese Journal of Tissue Engineering Research, 2013, 17(18): 3349-3357.
Quantitative analysis of experimental animals
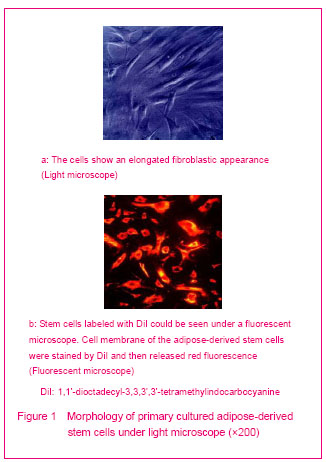
After primary cultured in the control medium, adipose-derived stem cells were harvested with an elongated fibroblast like appearance (Figure 1 a). After expanded to passages 3, the cells were detached and stained with fluorescent dye- 1,1'-dioctadecyl-3,3,3',3'-tetramethylindocarbocyanine (Figure 1 b). The 1,1’-dioctadecyl- 3,3,3',3'- tetramethylindocarbocyanine stained adipose-derived stem cells were selected for the experiment.
And then the hematoxylin-eosin staining showed that the neogenetic tissues were adipose tissues. The mean humid weight was (25.1±1.9) mg of the neogenetic tissue in the differentiated group was higher than that of the undifferentiated group (23.0±1.6) mg and the control group (20.2±1.4) mg (P < 0.01).

Vessel distribution observed with stereomicroscope
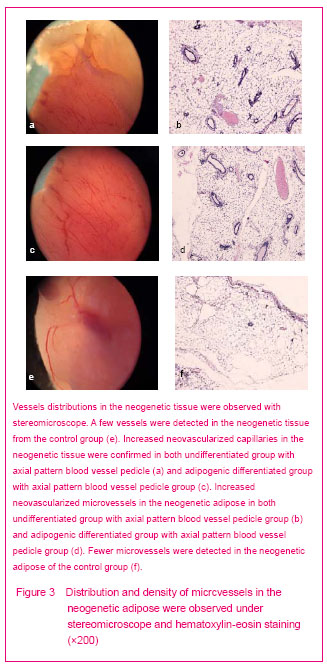
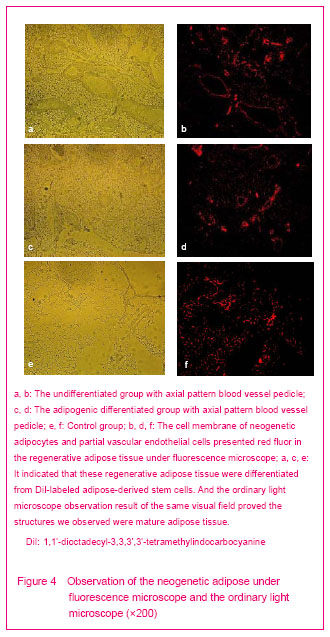
The adipocytes and partial vascular endothelial cells presented red fluor in the neogenetic tissue of all groups under fluorescence microscope (Figures 4b, d, f). It indicated that these cells were differentiated from DiI-labeled adipose-derived stem cells. The hematoxylin-eosin stained result proved the structure we observed was capillaries, and the scaffolds were degraded thoroughly (Figures 3b, d, f).
| [1] Asahara T, Masuda H, Takahashi T, et al. Bone marrow origin of endothelial progenitor cells responsible for postnatal vasculogenesis in physiological and pathological neovascularization. Circ Res. 1999;85(3):221-228.[2] Nakagami H, Maeda K, Morishita R, et al. Novel autologous cell therapy in ischemic limb disease through growth factor secretion by cultured adipose tissue-derived stromal cells. Arterioscler Thromb Vasc Biol. 2005;25(12): 2542-2547. [3] Planat-Benard V, Silvestre JS, Cousin B, et al. Plasticity of human adipose lineage cells toward endothelial cells: physiological and therapeutic perspectives. Circulation. 2004;109(5):656-663.[4] Boquest AC, Shahdadfar A, Brinchmann JE, et al. Isolation of stromal stem cells from human adipose tissue. Methods Mol Biol. 2006;325:35-46. [5] Boquest AC, Noer A, Collas P. Epigenetic programming of mesenchymal stem cells from human adipose tissue. Stem Cell Rev. 2006;2(4):319-329. [6] Kim Y, Kim H, Cho H, et al. Direct comparison of human mesenchymal stem cells derived from adipose tissues and bone marrow in mediating neovascularization in response to vascular ischemia. Cell Physiol Biochem. 2007;20(6): 867-876. [7] Lu F, Gao JH, Ogawa R, et al. Adipose tissues differentiated by adipose-derived stem cells harvested from transgenic mice. Chin J Traumatol. 2006;9(6):359-364. [8] Shi YY, Nacamuli RP, Salim A, et al. The osteogenic potential of adipose-derived mesenchymal cells is maintained with aging. Plast Reconstr Surg. 2005;116(6): 1686-1696. [9] Ogawa R, Mizuno H, Hyakusoku H, et al. Chondrogenic and osteogenic differentiation of adipose-derived stem cells isolated from GFP transgenic mice. J Nippon Med Sch. 2004;71(4):240-241. [10] Safford KM, Safford SD, Gimble JM, et al. Characterization of neuronal/glial differentiation of murine adipose-derived adult stromal cells. Exp Neurol. 2004;187(2):319-328. [11] Rodríguez LV, Alfonso Z, Zhang R, et al. Clonogenic multipotent stem cells in human adipose tissue differentiate into functional smooth muscle cells. Proc Natl Acad Sci U S A. 2006;103(32):12167-12172. [12] Mizuno H, Zuk PA, Zhu M, et al. Myogenic differentiation by human processed lipoaspirate cells Plast Reconstr Surg. 2002;109(1):199-209. [13] Planat-Benard V, Silvestre JS, Cousin B, et al. Plasticity of human adipose lineage cells toward endothelial cells physiological and therapeutic perspectives. Circulation. 2004;109(5):656-663. [14] Rehman J, Traktuev D, Li JL, et al. Secretion of angiogenic and antiapoptotic factors by human adipose stromal cells. Circulation. 2004;109(10):1292-1298. [15] Moon MH, Kim SY, Kim YJ, et al. Human adipose tissue-derived mesenchymal stem cells improve postnatal neovascularization in a mouse model of hindlimb ischemia. Cell Physiol Biochem. 2006;17(5-6):279-290. [16] De Ugarte DA, Alfonso Z, Zuk PA, et al. Differential expression of stem cell mobilization-associated molecules on multi-lineage cells from adipose tissue and bone marrow. Immunol Lett. 2003;31:267-270. [17] Ogawa R. The importance of adipose-derived stem cells and vascularized tissue regeneration in the field of tissue transplantation. Curr Stem Cell Res Ther. 2006;1(1): 13-20. [18] Friedenstein AJ. Precursor cells of mechanocytes. Int Rev Cytol. 1976;47:327-359. [19] Pittenger MF, Mackay AM, Beck SC, et al. Multilineage potential of adult human mesenchymal stem cells. Science. 1999;284(5411):143-147.[20] Reyes M, Lund T, Lenvik T, et al. Purification and ex vivo expansion of postnatal human marrow mesodermal progenitor cells. Blood. 2001;98(9):2615-625. [21] Jiang Y, Jahagirdar BN, Reinhardt RL, et al. Pluripotency of mesenchymal stem cells derived from adult marrow. Nature. 2002;418(6893):41-49. [22] Zuk PA, Zhu M, Mizuno H, et al. Multilineage cells from human adipose tissue: implications for cell-based therapies. Tissue Eng. 2001;7(2):221-228.[23] Terheyden H, Jepsen S, Rueger DR. Mandibular reconstruction in miniature pigs with prefabricated vascularized bone grafts using recombinant human osteogenic protein-1:a preliminary study. Int J Oral Maxillofac Surg. 1999;28(6):461-463. [24] Street J, Winter D, Wang JH, et al. Is human fracture hematoma inherently angiogenic? Clin Orthop. 2000;(378): 224-237.[25] MacLeod TM, Williams G, Sanders R, et al. Prefabricated skin flaps in a rat model based on a dermal replacement matrix Permacol. Br J Plast Surg. 2003;56(8):775-783. [26] Teng MS, Malkin BD, Urken ML. Prefabricated composite free flaps for tracheal reconstruction: a new technique. Ann Otol Rhinol Laryngol. 2005;114(11):822-826. [27] Tanaka Y, Sung KC, Fumimoto M, et al. Prefabricated engineered skin flap using an arteriovenous vascular bundle as a vascular carrier in rabbits. Plast Reconstr Surg. 2006;117(6):1860-1875. [28] The Ministry of Science and Technology of the People’s Republic of China. Guidance Suggestions for the Care and Use of Laboratory Animals. 2006-09-30. |
| [1] | 张同同, 王中华, 文 杰, 宋玉鑫, 刘 林. 3D打印模型在颈椎肿瘤手术切除与重建中的应用[J]. 中国组织工程研究, 2021, 25(9): 1335-1339. |
| [2] | 吴 训, 孟娟红, 张建运, 王 亮. 浓缩生长因子修复兔髁突全层软骨损伤[J]. 中国组织工程研究, 2021, 25(8): 1166-1171. |
| [3] | 曾燕华, 郝延磊. 许旺细胞体外培养及纯化的系统性综述[J]. 中国组织工程研究, 2021, 25(7): 1135-1141. |
| [4] | 刘 聪, 刘 肃. miR-17-5p调控低氧诱导因子1α介导脂肪细胞分化及血管生成的分子机制[J]. 中国组织工程研究, 2021, 25(7): 1069-1074. |
| [5] | 杨 鑫, 金 喆, 冯 旭, 卢 兵. 沈阳市居民对器官、眼组织及遗体捐献的认知及意愿调查[J]. 中国组织工程研究, 2021, 25(5): 779-784. |
| [6] | 徐东紫, 张 婷, 欧阳昭连. 心脏组织工程领域全球专利竞争态势分析[J]. 中国组织工程研究, 2021, 25(5): 807-812. |
| [7] | 吴子健, 胡昭端, 谢有琼, 王 峰, 李 佳, 李柏村, 蔡国伟, 彭 锐. 3D打印技术与骨组织工程研究文献计量及研究热点可视化分析[J]. 中国组织工程研究, 2021, 25(4): 564-569. |
| [8] | 常文辽, 赵 杰, 孙晓亮, 王 锟, 吴国锋, 周 剑, 李树祥, 孙 晗. 人工骨膜的材料选择、理论设计及生物仿生功能[J]. 中国组织工程研究, 2021, 25(4): 600-606. |
| [9] | 刘 旒, 周箐竹, 龚 桌, 刘博言, 杨 斌, 赵 娴. 胶原/无机材料构建组织工程骨的特点及制造技术[J]. 中国组织工程研究, 2021, 25(4): 607-613. |
| [10] | 刘 飞, 崔宇韬, 刘 贺. 局部抗生素递送系统治疗骨髓炎的优势与问题[J]. 中国组织工程研究, 2021, 25(4): 614-620. |
| [11] | 李晓壮, 段 浩, 王伟舟, 唐志宏, 王旸昊, 何 飞. 骨组织工程材料治疗骨缺损疾病在体内实验中的应用[J]. 中国组织工程研究, 2021, 25(4): 626-631. |
| [12] | 张振坤, 李 喆, 李 亚, 王莹莹, 王亚苹, 周馨魁, 马珊珊, 关方霞. 海藻酸盐基水凝胶/敷料在创面愈合中的应用:持续、动态与顺序释放[J]. 中国组织工程研究, 2021, 25(4): 638-643. |
| [13] | 陈佳娜, 邱燕玲, 聂敏海, 刘旭倩. 组织工程支架材料修复口腔颌面部软组织缺损[J]. 中国组织工程研究, 2021, 25(4): 644-650. |
| [14] | 邢 浩, 张永红, 王 栋. 长骨大段骨缺损修复方法的优势与不足[J]. 中国组织工程研究, 2021, 25(3): 426-430. |
| [15] | 高 坤, 陈大宇, 张 勇, 刘伟东, 孙淑芬, 赖文强, 马笃军, 吴益宏, 林展鹏, 蒋鹰鹭, 余伟吉. 牛膝醇提物调控滑膜成纤维细胞外泌体抑制软骨细胞外基质降解[J]. 中国组织工程研究, 2021, 25(23): 3636-3640. |
Adipose-derived stem cells are a feasible cell source for adipose tissue regeneration. However, the capital task is how to enhance the adipogenic conversion of adipose-derived stem cells in vivo. In vitro, the adipogenic differentiation of adipose-derived stem cells prior to implantation might enhance the adipose tissue regeneration. Yet, the ability of adipogenesis when adipogenic-induced adipose-derived stem cells implanted is different site to site. The fascia musculares around blood vessel pedicle of latissimus dorsi myocutaneous flap is characterized by abundant blood supply. So the compounds of adipose-derived stem cells-loaded honeycomb collagen scaffolds would obtain adequate nutrient blood flow from the pedicle of latissimus dorsi myocutaneous flap when the cells were transplanted into there.
Design
.jpg)
1 实验成功构建了带血管肌筋膜包埋脂肪干细胞载体复合物构建血管化组织工程脂肪,成脂效率较好。 2 文章结果证实,将脂肪干细胞载体复合物包埋在带血管蒂肌筋膜包埋中能促进脂肪干细胞的成活及组织工程脂肪的脂肪化,并且证明了自体脂肪干细胞能够分化为血管内皮细胞进而促进血管的新生。 基金项目: 课题受中国博士后科学基金(20090450910),广东省医学科研基金项目(A2011739)和中山市科技计划项目(20113A008)资助。
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||